Explain the long form of the periodic table.How does the number of valence electrons vary on moving from left to right:(i) In the first period of the periodic table? (ii) In the second period of the periodic table?.Thus, the periodic table becomes a tool for remembering the charges on many ions. Consider the following elements:Na, Ca, Al, K, Mg, Li (a) Which of these elements belong to the same period of the periodic table? (b) Which of these elements belong to the same group of the periodic table? In many cases, elements that belong to the same group (vertical column) on the periodic table form ions with the same charge because they have the same number of valence electrons.(a) What is the main characteristic of the last elements in the periods of the periodic table? What is the general name of such elements?(b) What is the number of elements in: (a) 1st period, and (b) 3rd period, of the modern periodic table?.
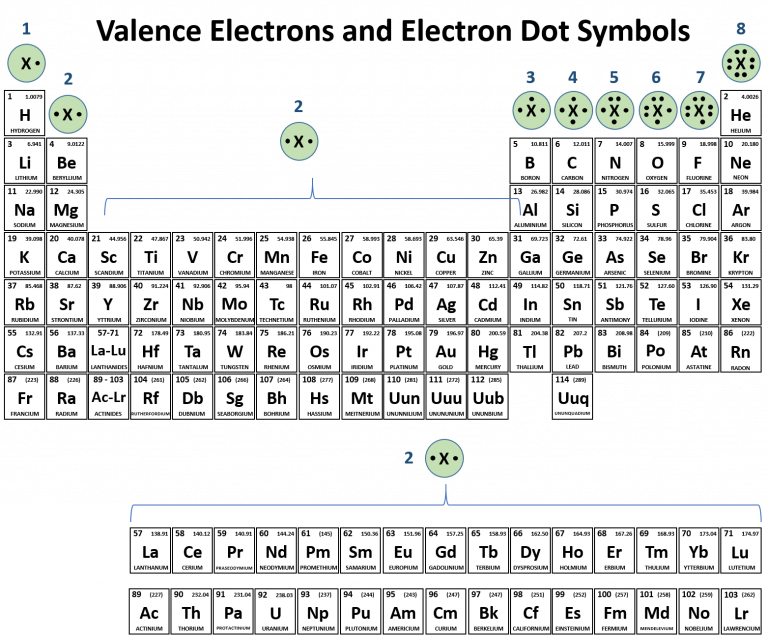
There are some exceptions to the predicted filling order, particularly when half-filled or completely. In the periodic table, elements with analogous valence electron configurations usually occur within the same group. The Shorthand electron configuration (or Noble gas configuration) as well as Full electron configuration is also mentioned in the table. Electrons in the outermost orbitals, called valence electrons, are responsible for most of the chemical behavior of elements.

(a) What is the usual number of valence electrons and valency of group 18 elements of the periodic table?(b) What happens to the number of valence electrons in the atoms of elements as we go down in a group of the periodic table?.(a) How does the electropositive character of elements change on going down in a group of the periodic table?(b) State how the valency of elements varies (i) in a group, and (ii) in a period, of the periodic table.The periodic table and trends in valence electrons can be used to determine the number of bonds an atom is most likely to form. Atoms share electrons and form covalent bonds to satisfy the octet rule. In the modern periodic table, which are the metals among the first ten elements? 4.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts.Compare and contrast the arrangement of elements in Mendeleev’s Periodic Table and the Modern Periodic Table.What is the major characteristic of the first elements in the periods of the periodic table? What is the general name of such elements?.Name of 118 elements in the periodic table.How does the valency of elements change on moving from left to right in the third period of the periodic table?.
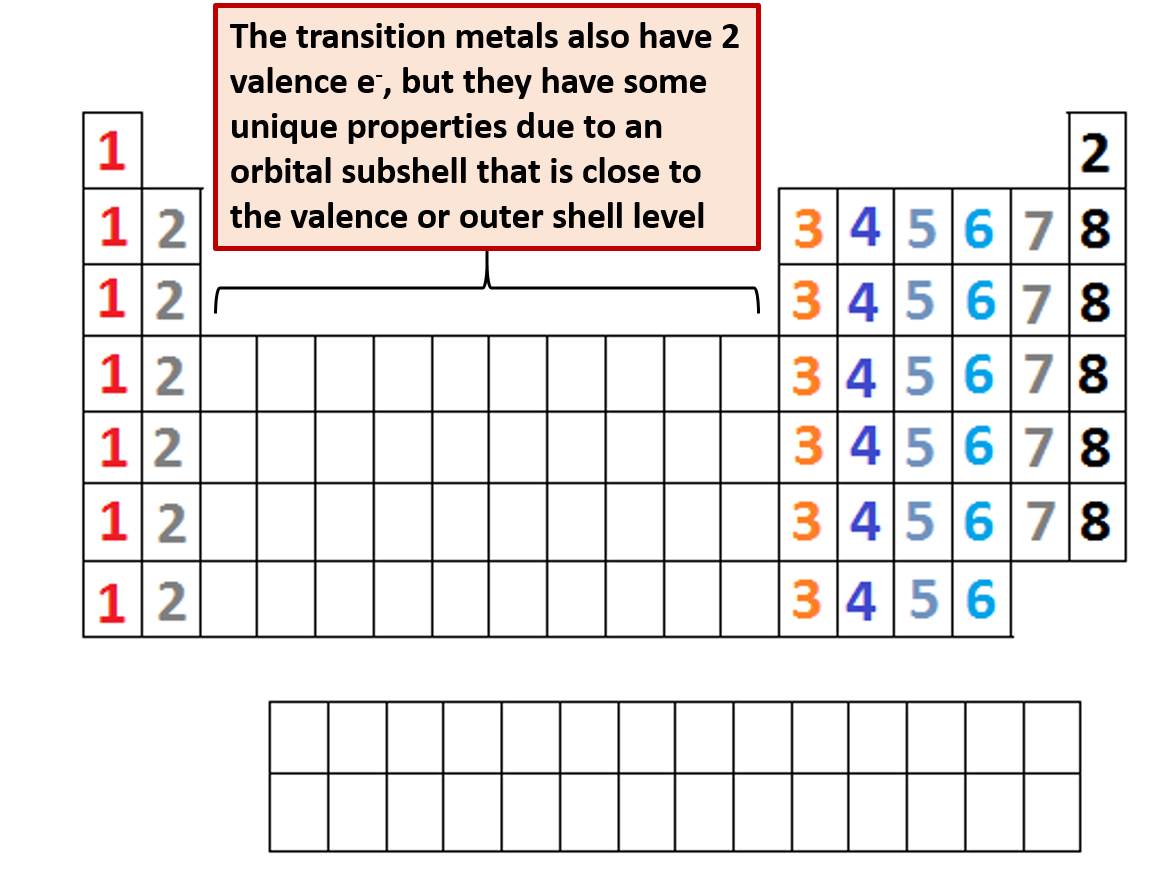
How does the valency of elements vary in going down a group of the periodic table?.

